CelCradle X® Closed Automated Single-Use Bioreactor
The Next Cradle Generation for Adherent Culture


The cyclic upward and downward motion, or Tide Motion in CelCradle X provides alternate exposure of nutrition and aeration to the cells attached packed bed. The packed bed varies from BioNOC™ II, BioMESH® macroporous carriers or the user’s own.
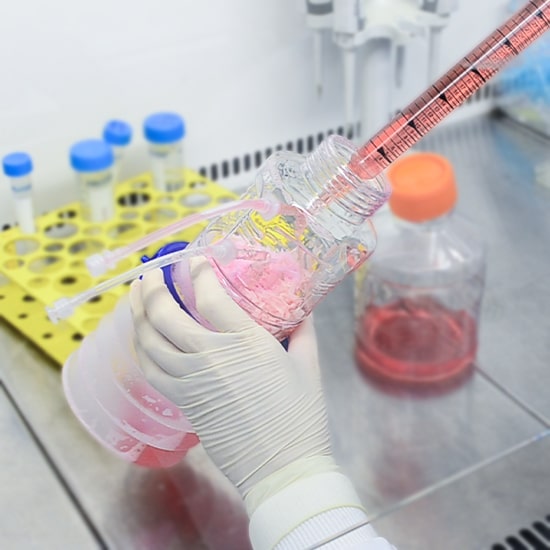

Automated single-use bioreactor for autologous cell therapy has becoming a modality in the industry. Aside from its advantages for its straightforward approach in culture, it provides a more robust approach to eliminate or reduce obstacles faced in manufacturing. CelCradle X® (CCX) is a combination of both single-use or multiple use mixer for achieving a safe and effective adherent culture production.
The CCX-SA-SUGH is a semi-automated bioreactor model that uses a single-use CCX bottle and a glass or single-use mixer. The mixer component varies from the process mode needed in the culture run. For processes that require perfusion mode, a 0.5L single-use vessel shall be used to accommodate the embedded feeding and harvesting rate. Sensors can either be autoclavable or inline depending on the user’s requirement.
The CCX-SA-SUGH that opt to do a batch process, the 2 L glass vessel shall be used. Base parameters such as maintaining glucose concentration as well as pH will be controlled during the culture run.
The CCX-SA-SUB model is a semi-automated bioreactor that only uses single-use components from its main vessel (where the packed bed is) to mixer (for culture medium homogeneity). The pH/DO sensors are also single-use thus offering a complete single-use solution for the adherent culture process. For perfusion processes required in the run, a 0.5 L single-use vessel equipped with in line sensors shall be used. The main vessel is packed with a 0.1 L BioNOC™ II or BioMESH® for cell adherence in 3D.
Batch process includes a single-use main vessel packed with 0.1 L BioNOC™ II or BioMESH® with a 2 L single-use mixer. This model is equipped with separate single-use bags for glucose maintenance and base solutions.
The CelCradle X single-use bioreactor helps to provide solutions faced in adherent culture processes especially in its expansion, viral transfection/transductions, and more. The production of stem cells for example in a 0.1 L packed bed offers up to 6,400 cm2 surface area for 3D cell growth. This in vivo-like nature of macroporous carriers maintains the cell’s characteristics and stable cell culture environment either in hypoxic or normoxic conditions.
Manual processes are reduced in this format as CCX can linearly scale-up to TideXcell® bioreactor especially when switching to allogeneic cell therapy. The CCX can automatically harvest cells using the CCX®-SAH with more than 90% efficiency. It also reduced the need for GMP-grade cleanrooms and consumable costs.
Sign up to our newsletter and receive the latest news and updates about our products!
