Applications
MDCK cell lines & Vero cell lines
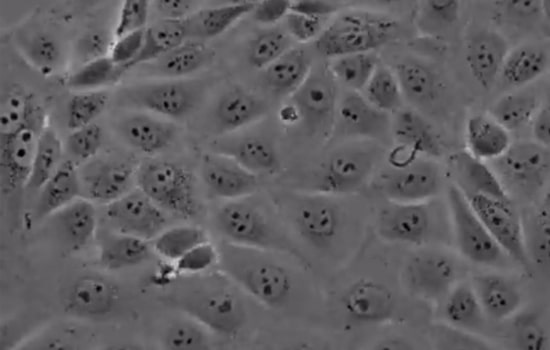
MDCK cell lines
Adherent mammalian cell lines such as MDCK which can be grown in serum-free medium are currently the most popular as compared to Vero and have been reported to induce higher hemagglutination inhibition (HI) and neutralizing antibody (NAb) titers than egg-based vaccine. Particularly, notable is that adherent MDCK outperform the suspension form by producing a more robust virus.
Other specific advantages of MDCK lines are enabling rapid replication of virus in 3-10 passages (10-30d) resulting in high-titer viruses with fewer mutations. The net result is a shortening of the lead time for Vaccine production. Influenza B is particularly suitable for propagation in these cell lines. The safety and immunogenicity of vaccines from MDCK lines have been well-documented.
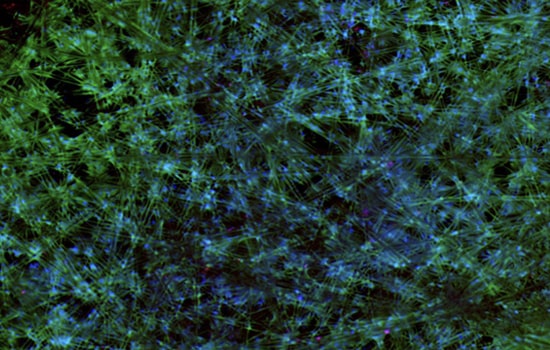
Vero cell lines
Lines of investigation support the Vero cell line as the candidate of choice for viral vaccine production, including: efficiency of primary virus isolation and replication to high infectivity titers; genetic stability of the hemagglutinin molecule, while maintaining the antigenic properties of human-derived viruses; and similarities in the pattern of protein synthesis and morphological changes between virus-infected Vero and MDCK cells. Additionally, Vero is the only cell line that has received worldwide regulatory acceptance for several vaccines.
In the case of H5N1, WHO requires BSL 3 to produce a Vaccine. The highest biosafety level available in egg-based vaccine manufacturing is BSL 2. The generation and safety testing of the procedure may take weeks and will result in the delay in the delivery of the vaccine. Cell-based production solves this issue as VERO cell cultures allow high-titer growth of the said strain under BSL 3 conditions.





