What is Poliomyelitis?
Poliomyelitis also known as Polio or infantile paralysis, is a highly contagious viral infection caused by poliovirus. It invades the nervous system and can cause irreversible paralysis, breathing problems and even death. It is caused by one of the three polioviruses from genus Enterovirus: Human poliovirus 1 (HPV1), Human poliovirus 2 (HPV2) and Human poliovirus 3 (HPV 3). Polio can strike at any age, but mainly affects children under 5 years old.
Transmission

Poliovirus is transmitted from one person to another through oropharyngeal secretions and fecal contamination. An infected person may spread the virus immediately for about one to two weeks after the symptoms appear. The virus will enter the body through the mouth by oropharyngeal secretion and will multiply in the intestines then spread through the feces. The virus is intermittently excreted one month or more after infection.
Signs and Symptoms

Polio is a silent disease that progresses overtime. The initial signs are fever, muscle pain, vomiting and nausea. It may lead to temporary or permanent paralysis when left undiagnosed and not prevented. Most infected are not aware that they already have poliovirus.
Up to this day, there is no certain cure for patients infected with poliovirus. For this reason, vaccines are extremely important to prevent infection from occurring.
The Poliomyelitis Vaccines
|
Oral Polio Vaccine (OPV) |
Inactivated Polio Virus (IPV) |
|
|
|
|
|
|
|
Trivalent oral poliovirus vaccine (tOPV): Protects against types 1, 2 and 3 Bivalent oral poliovirus vaccine (bOPV): Protects against type 1 and 3 Monovalent oral poliovirus vaccine (mOPV): Protects either type 1, 2 or 3 |
IPV: protects against types 1, 2 and 3 |
Polio Eradication
Immunization efforts have reduced the number of polio cases globally by more than 99%. During 1999, out of the three types of poliovirus (types 1, 2 and 3), the type 2 wild poliovirus has been successfully eradicated.
|
2014-2015 |
April 2016 |
2019-2020 |
|
Introduction to IPV |
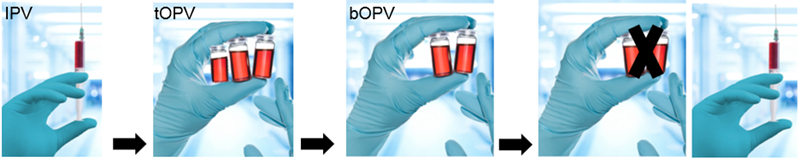
Switch from tOPV to bOPV |
Withdrawal of OPV vaccine |
Why switch tOPV to bOPV?
The three types of wild poliovirus (Type 1, 2 and 3) is targeted by different component of the tOPV.
Live attenuated vaccines are effective against the poliovirus, but in extremely rare conditions it can lead to paralysis. There are two ways this can take place:
Although wild poliovirus type 2 is successfully eradicated globally since 1999, vaccine-related type 2 viruses continue to cause the majority of cVDPV outbreaks and many VAPP cases.
Therefore, OPV type 2 today transfers more risk than benefit and declines global polio eradication efforts. Thus, tOPV will be replaced with bOPV, which will continue to target the remaining polio types 1 and 3. When these types are eradicated, bOPV will also be withdrawn.
VacciXcell Solution
Poliovirus is a non-secreted virus which reproduce within the cultured cells. Since the virus are contained within host cells, the cells themselves must be harvested and processed in order to get the viruses. After the cells are harvested, methods such as freeze and thaw is used to lyse cells and harvest the virus.
In order to make the vaccine, huge amount of poliovirus must be processed. Poliovirus are commonly proliferated by using Vero cell line as host cells.
VacciXcell's tide motion bioreactor is the most effective and innovative equipment for high density cell culture. It is perfect for non-secreted virus production due to two major fact, its proprietary BioNOC II macrocarrier and its proprietary tide Motion technology.
Tide Motion Bioreactors

CelCradle™
CelCradle™ is a single use bioreactor capable of high-density adherent cell culture for virus production. It is capable of producing Vero cells in high density. This provides a low shear stress, high aeration, and foam-free culture environment.

BioNoc II
BioNOC II macrocarriers ensures that the cells are fixed during and after culture, making sure that there are no cells that will be harvested along with the media, immediately lessening the culture broth content thus reducing downstream processing requirements.

VacciXcell™ Hybrid Bioreactor
Four-in-One - VacciXcell™ Hybrid Bioreactor is capable of performing four (4) types of culture processes namely adherent culture using micro carrier, adherent culture using macro carrier, suspension culture and fermentation. Adherent culture is performed through patented tide motion technology while suspension culture and fermentation are performed through standard stirred tank process.
Perfect for Virus Production using adherent cell lines
Perfect for Vaccine Research and Development when deciding between suspension or adherent cell lines or converting adherent cell lines to suspension, data can be easily compared. Comparison can be made by using conventional micro carriers or our packed bed BioNoc II (when using tide motion).

TideCell®
100% nutrient exchange for highest virus titer - Media used for cell growth can be 100% pumped out as waste and 100 % new media is used after virus seeding for virus growth.
When culturing difficult viruses, parameters such as temperature, pH and oxygen levels are automatically monitored and adjusted to cater to specific virus requirements. TideCell™ also has zero shear stress for stress sensitive viruses and is capable of harvesting using an automated harvesting system for non-secreted viruses.
It is the pilot and production scale system of Esco VacciXcell’s Tide Motion Bioreactors.
Sign up to our newsletter and receive the latest news and updates about our products!
